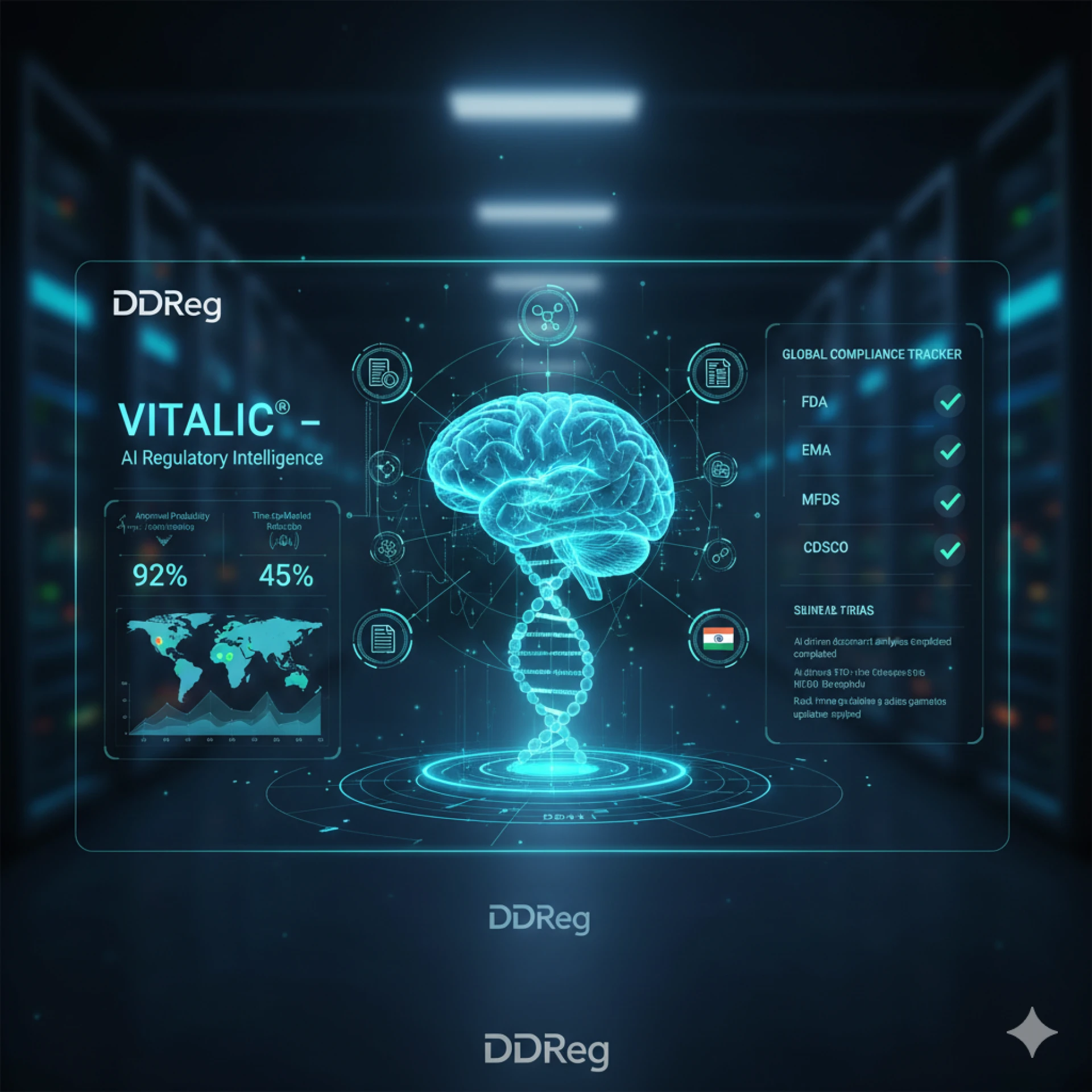
Unlocking Regulatory Foresight: How DDReg's RegIntel Transforms Your Global Compliance Strategy
The pharmaceutical industry operates within an ever-shifting sea of regulations. For companies striving for global marke

DDReg Pharma is a trusted global partner in the life sciences industry, delivering specialized consulting solutions across Regulatory Affairs, Pharmacovigilance, Quality, IPR, GMP Compliance, and Toxicology.
With over 15 years of experience, we help pharmaceutical, and biopharma companies accelerate product registrations and ensure end-to-end regulatory compliance — from early-stage development to post-marketing lifecycle management.
Backed by in-house tools like our AI-driven Regulatory Intelligence Platform and Regulatory Information Management System (RIMS), we enable smarter, faster, and more compliant regulatory operations.

The pharmaceutical industry operates within an ever-shifting sea of regulations. For companies striving for global marke

The journey of a medicine from the laboratory to the patient is long and rigorous. Clinical trials meticulously evaluate

A dossier in pharma is a comprehensive and highly structured collection of documents that a pharmaceutical company submi

In the ever-evolving world of medical devices, regulatory compliance is not just a necessity; it is a critical part of e

Navigating the Labyrinth: Regulatory Services for Pharmaceutical Products in IndiaIndia's pharmaceutical industry is a g
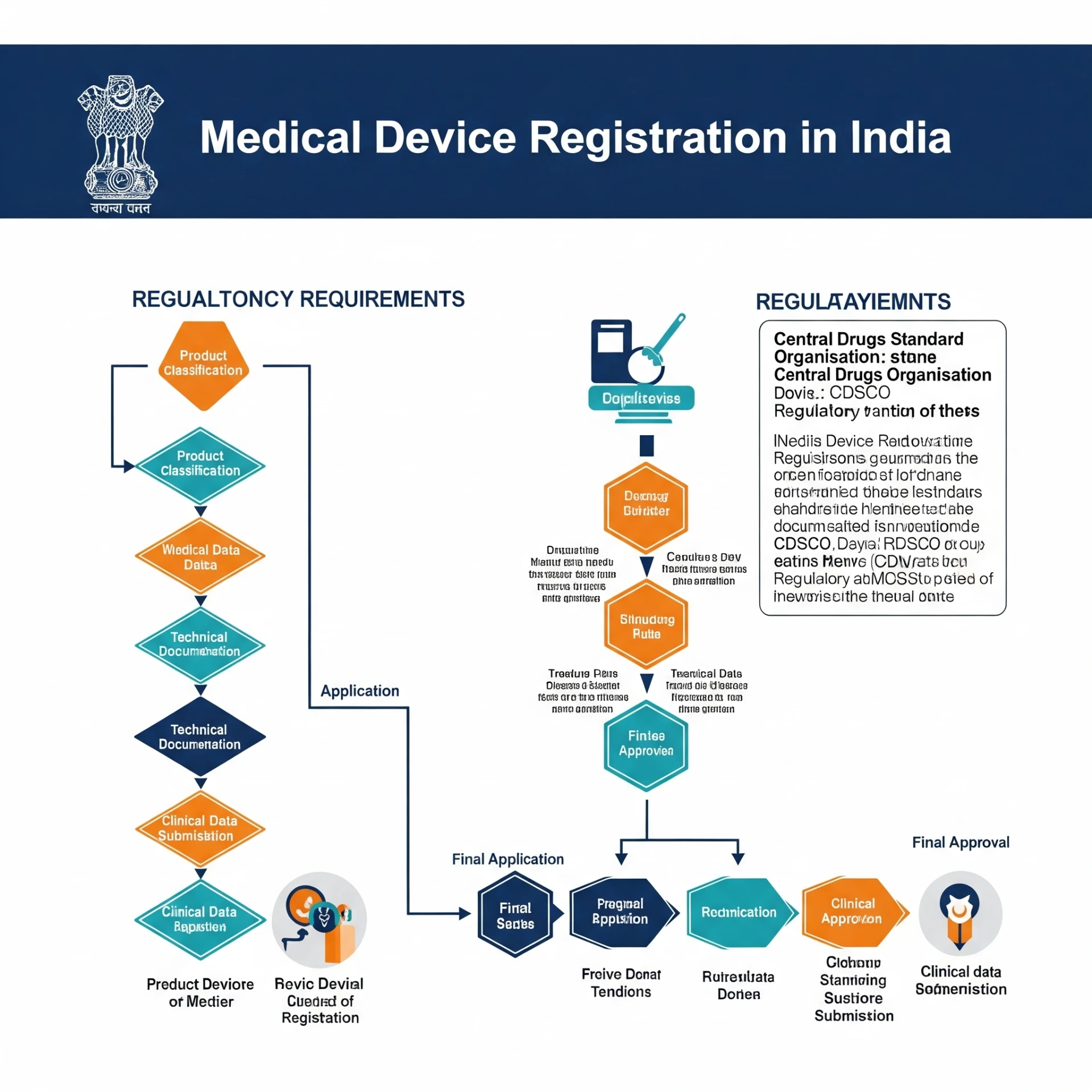
Navigating the Indian Market: Medical Device Registration in IndiaIndia's healthcare sector is experiencing rapid growth

Clinical Research Services in India: A Growing Hub for Global TrialsIndia has emerged as a significant destination for C
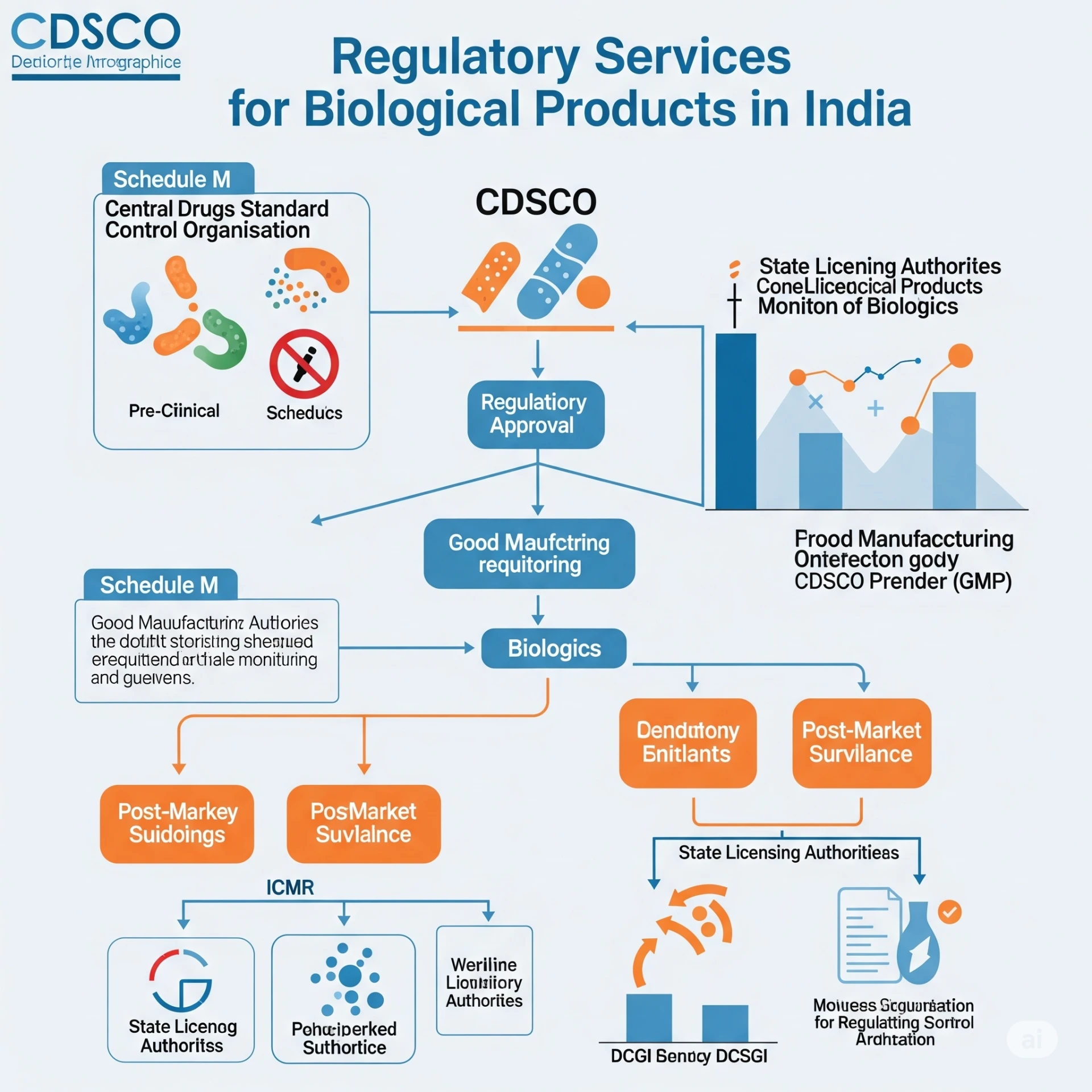
Regulatory Services For biological Product in IndiaIndia has rapidly emerged as a significant player in the global pharm

India’s rapidly expanding healthcare market presents immense opportunities, but also a complex regulatory environment. F

Pharmaceutical companies face complex challenges in obtaining product approvals, managing compliance across various stag

Data Privacy Regulations in Global Clinical Trials: What You Need to Know

Navigating the Complexities of Risk Assessment in Veterinary Pharmaceutical